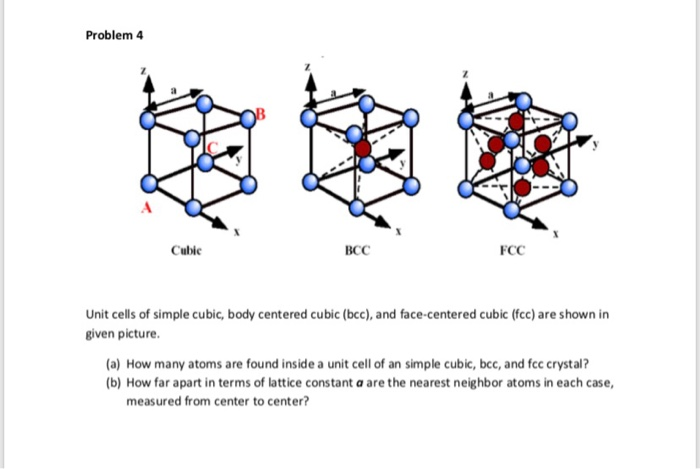
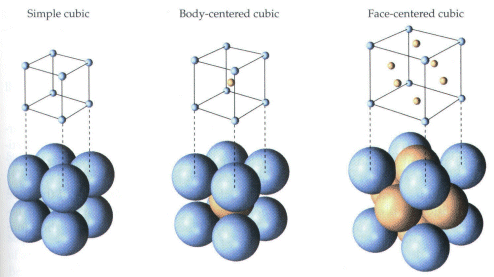
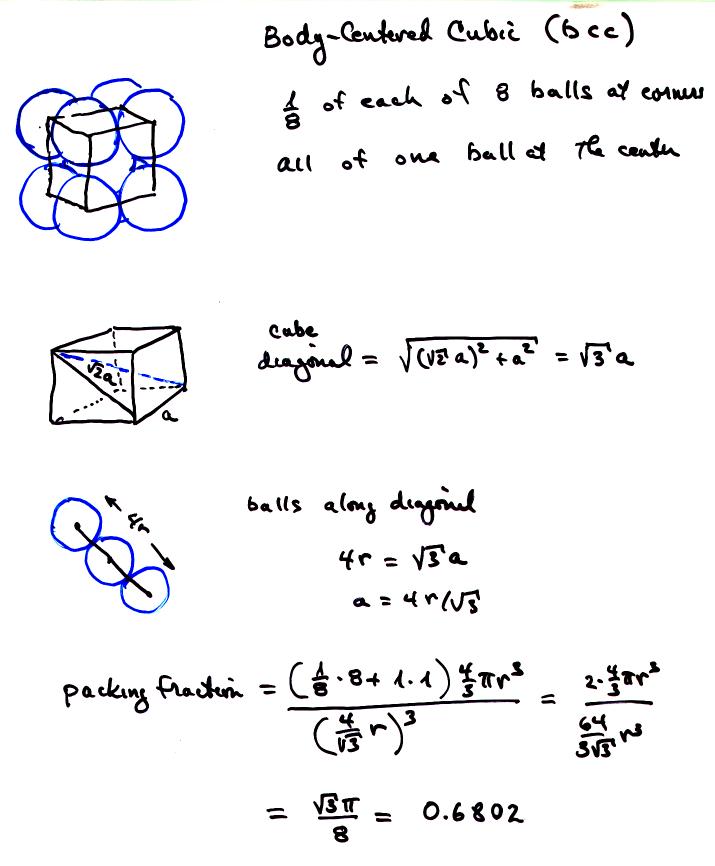
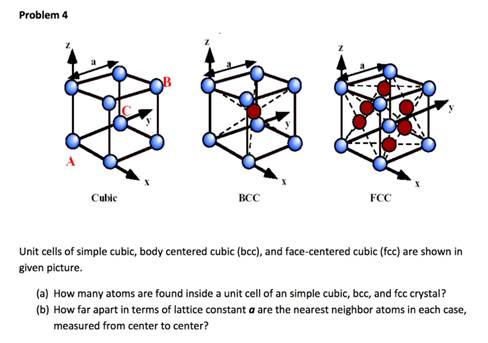
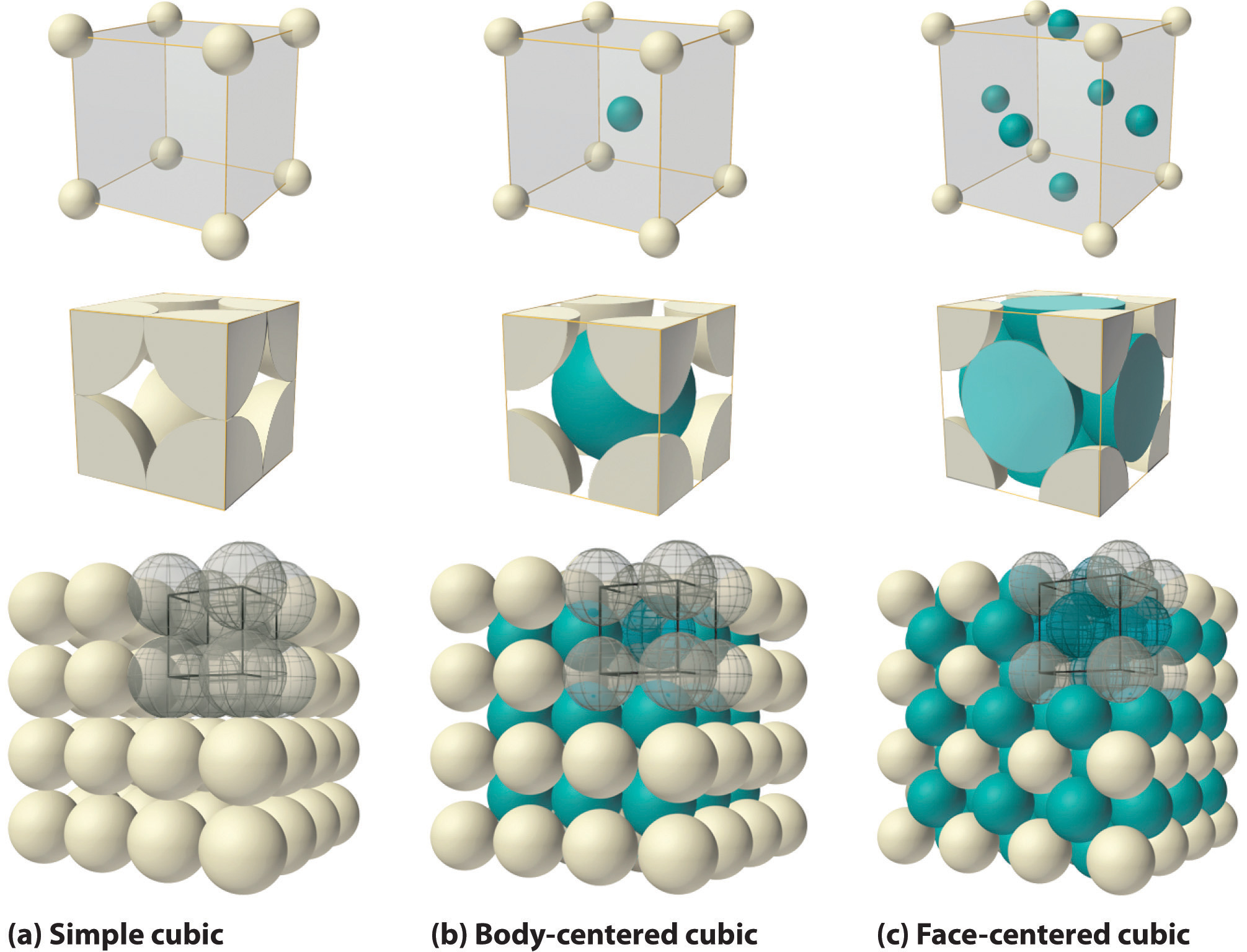
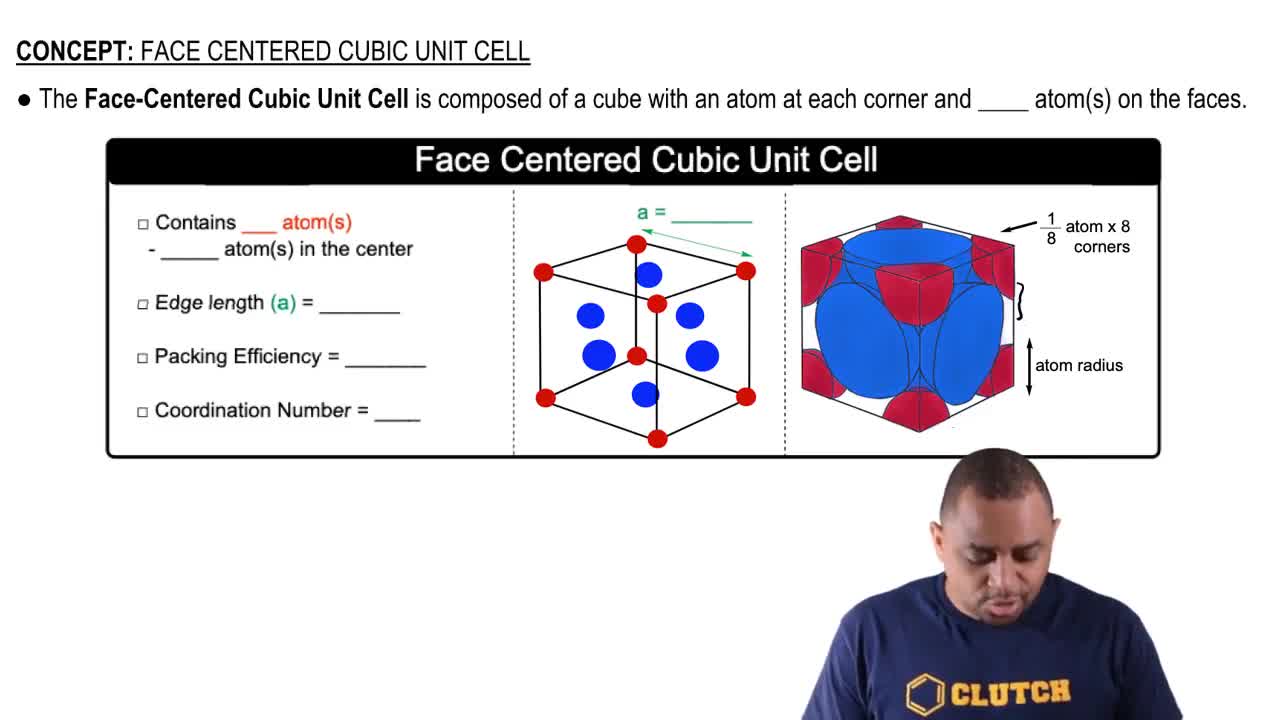
all Souad BD PROBLEM II Silver crystallises in face centred cubic structure. The edge length of unit cell is found to be 108.7 pm. Calculate density of silver (Ag=108g mol') Solution Edge

An element has a body-centered cubic (bcc) structure with a cell edge of 288pm. The density...... - YouTube
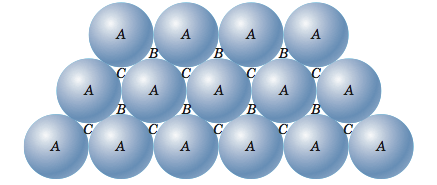
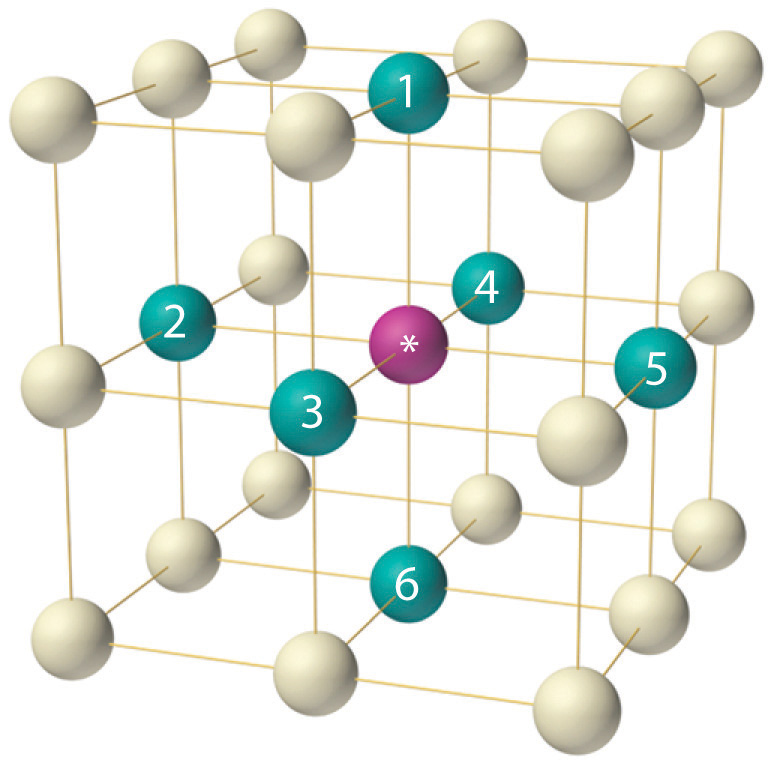
47.What is the number of second nearest neighbour in simple cubic unit cell, body center cubic unit cell, face centered cubic unit cell.